Reciprocal restrictions will be applied on EU countries barring Indian entry, say sources
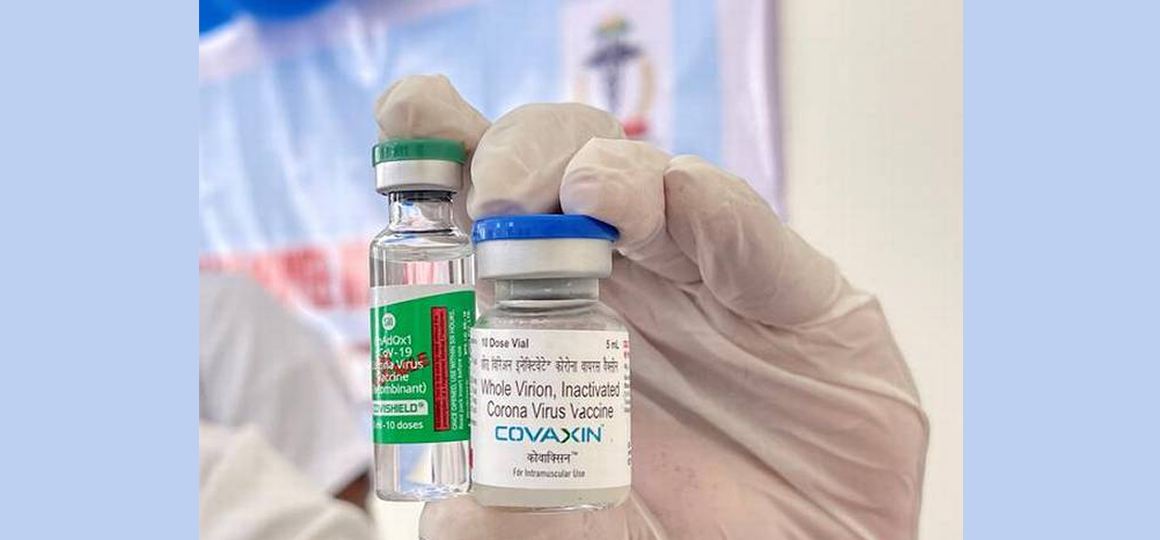
India took issue with the European Union’s plans to institute a “green pass” from July 1, with government sources warning that India will introduce a “reciprocal policy” only allowing ease of travel for those European countries that recognise Indian vaccines Covishield and Covaxin.
The latest twist to the controversy of the European Union’s Digital Covid Certificate came a day after External Affairs Minister S. Jaishankar met with his EU counterpart High Representative Josep Borrell Fontelles. In a tweet after the meeting, Mr. Jaishankar indicated that he raised the issue of possible restrictions on Indian travellers in the EU if they don’t have one of four vaccines approved by the European Medicines Agency (EMA), that include the Europe made Astra Zeneca vaccine but not the Indian made Serum Institute of India’s (SII) Covishield or Bharat Biotech’s Covaxin.
“We have requested EU Member States to individually consider extending similar exemption to those persons who have taken COVID-19 vaccines in India, that is, Covishield and Covaxin, and accept the vaccination certificate issued through the CoWIN portal,” the sources said on Wednesday.
“We have also conveyed to EU Member States that India will institute a reciprocal policy for recognition of the EU Digital Covid Certificate,” the sources added, making it clear that only citizens of those countries in the 27-member grouping who make exemptions for travellers inoculated with the Indian vaccines would be given an exemption from mandatory quarantine on arrival in India.
The government’s statement, made through officials who didn’t wish to be identified, came even though the European Union had issued a detailed clarification, denying its plan was discriminatory. The African Union had also issued a letter of protest over the EU proposal saying that it promoted “inequality” for those from India and “lower-income” countries for whom the Covishield vaccine was the “backbone” of the international COVAX alliance programme.
The EU had also explained that SII had not applied for the requisite permissions with the EMA, and had promised to consider its case as soon as it applied. However, New Delhi now appears to be taking the position that the EMA must consider the Indian-made vaccine by accepting the CoWin certificate, regardless of the applications.
The government’s stand is especially significant since while Covishield has received World Health Organization authorisation, Covaxin is yet to be cleared by the global health body.
India toughens stand on EU COVID passport
Reciprocal restrictions will be applied on EU countries barring Indian entry, say sources
India took issue with the European Union’s plans to institute a “green pass” from July 1, with government sources warning that India will introduce a “reciprocal policy” only allowing ease of travel for those European countries that recognise Indian vaccines Covishield and Covaxin.
The latest twist to the controversy of the European Union’s Digital Covid Certificate came a day after External Affairs Minister S. Jaishankar met with his EU counterpart High Representative Josep Borrell Fontelles. In a tweet after the meeting, Mr. Jaishankar indicated that he raised the issue of possible restrictions on Indian travellers in the EU if they don’t have one of four vaccines approved by the European Medicines Agency (EMA), that include the Europe made Astra Zeneca vaccine but not the Indian made Serum Institute of India’s (SII) Covishield or Bharat Biotech’s Covaxin.
“We have requested EU Member States to individually consider extending similar exemption to those persons who have taken COVID-19 vaccines in India, that is, Covishield and Covaxin, and accept the vaccination certificate issued through the CoWIN portal,” the sources said on Wednesday.
“We have also conveyed to EU Member States that India will institute a reciprocal policy for recognition of the EU Digital Covid Certificate,” the sources added, making it clear that only citizens of those countries in the 27-member grouping who make exemptions for travellers inoculated with the Indian vaccines would be given an exemption from mandatory quarantine on arrival in India.
The government’s statement, made through officials who didn’t wish to be identified, came even though the European Union had issued a detailed clarification, denying its plan was discriminatory. The African Union had also issued a letter of protest over the EU proposal saying that it promoted “inequality” for those from India and “lower-income” countries for whom the Covishield vaccine was the “backbone” of the international COVAX alliance programme.
The EU had also explained that SII had not applied for the requisite permissions with the EMA, and had promised to consider its case as soon as it applied. However, New Delhi now appears to be taking the position that the EMA must consider the Indian-made vaccine by accepting the CoWin certificate, regardless of the applications.
The government’s stand is especially significant since while Covishield has received World Health Organization authorisation, Covaxin is yet to be cleared by the global health body.




NO COMMENT